
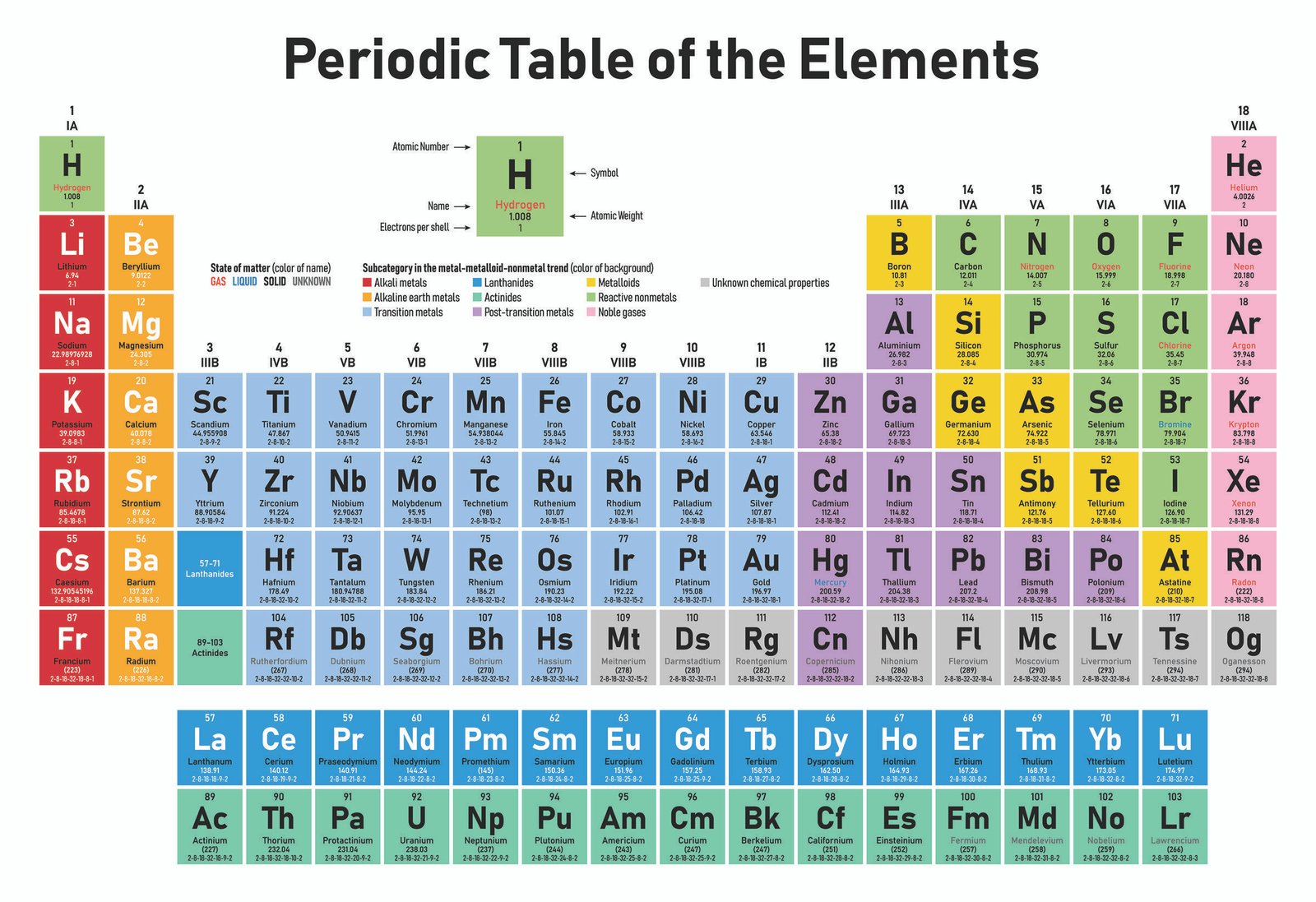
We now know what the periodic table looks like and how it is structured. They are average conductors of electricity.They have medium electronegativity values.Typically, metalloids are shiny and lustrous when cut, but are brittle in nature.They straddle the dividing line that splits metals from non-metals, and their properties are halfway between the two. Metalloids are found in the middle of the periodic table. 
They're poor conductors of heat and electricity.Some, like silicon, have extremely high melting points whilst others, like oxygen, have low melting points. They exhibit a range of melting and boiling points.They have high electronegativity values.Non-metals typically gain electrons to form negative anions.In fact, you can think of them as simply lacking in metallic characteristics. 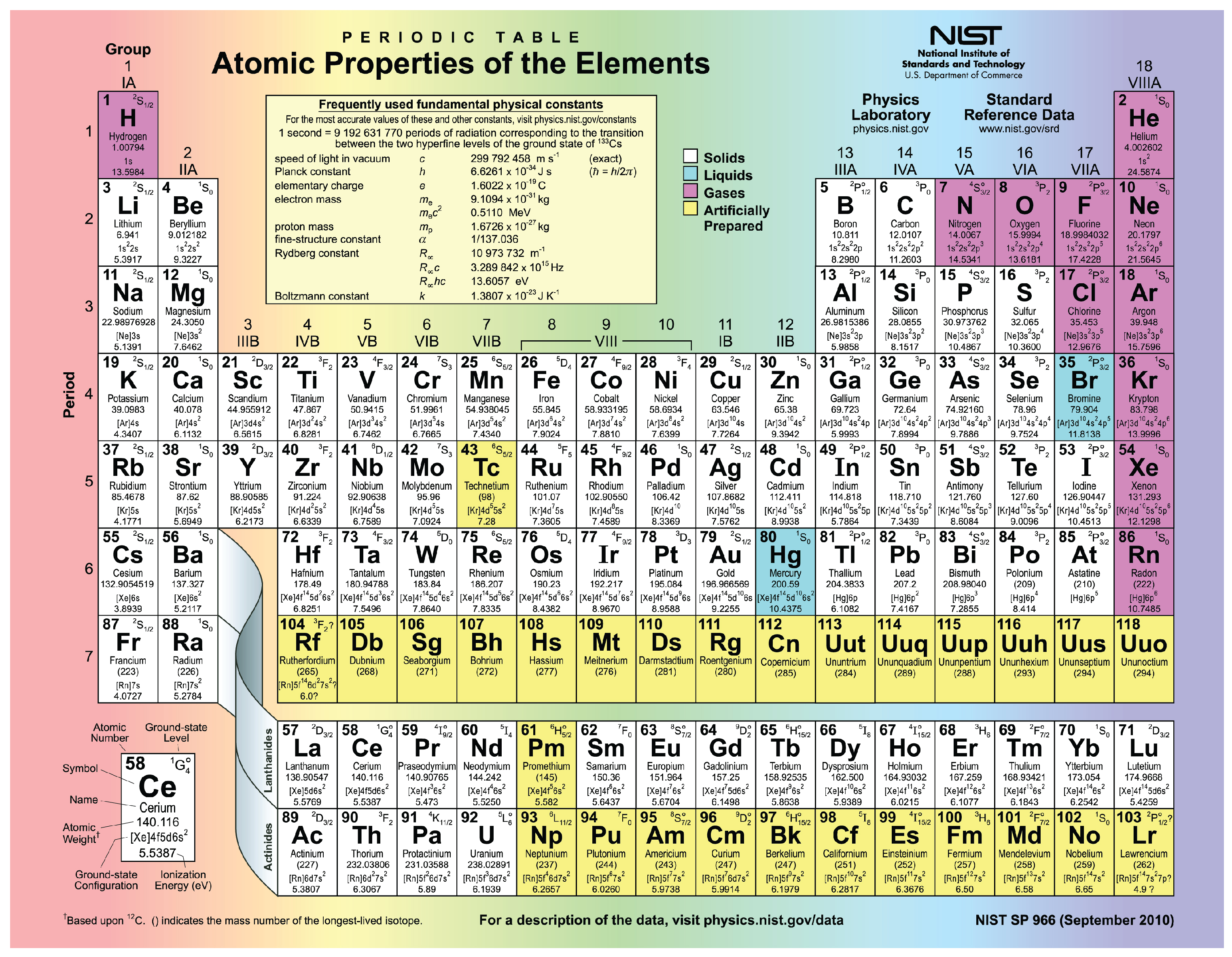
As the name suggests, they're the opposites of metals. Non-metals are found on the right-hand side of the periodic table (with the exception of hydrogen, which is also a non-metal).
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
